News

MEDICA Trade Fair 2023
Visit us at MEDICA 2023
The live event will be held from November 13 - 16, 2023 and we will be there.
We are exhibiting together with our mother company, BioVendor, in Hall 3 A92.
We are really looking forward to meet with you again in person!

NGS Contract Award Phase 2
ViennaLab Diagnostics and its partners secured funding for phase 2 of the prestigious oncNGS project.
ViennaLab Diagnostics partnered together with Euformatics and Oncompass Medicine and progressed to the phase 2 to design a state-of-the-art solution which aims to revolutionize the field of liquid biopsy and cancer detection.
Phase 2 will last 12 months, during which, we will apply our expertise to develop a non-invasive Pan-cancer cfDNA test to profile the tumor landscape and predict the best personalized treatment for every patient.
For more information, follow the link to Media Centre announcement. Please click here for the official Press Release.

First IVDR-compliant products!
The EU IVDR 2017/746 directive, commonly referred to as IVDR, provides high levels of patient safety and quality while promoting innovation.
To avoid shortages of in-vitro diagnostic products at service laboratories when the IVDR directive is enforced, ViennaLab has initiated the transition of its products to IVDR by submitting the technical documentation for the first tranche of our RealFast™ Assays and StripAssays® to the Notified Body TÜV Süd and successfully obtaining approval.
This means that we now offer you the following products developed and produced under IVDR:
- alpha-Globin StripAssay [REF 4-160]
- CAH StripAssay [REF 4-380]
- CF StripAssays [REF 4-410, 4-420, 4-430, 4-440]
- FMF StripAssay [4-230]
- FMF-SAA1 StripAssay [REF 4-390]
- HLA-B27 RealFast Assay [REF 7-620/7-623]
By using ViennaLab assays in your diagnostic laboratory, you save valuable time and resources. There is no need to create technical documentation, demonstrate scientific validity, or generate analytical, clinical, and performance data—tasks that are both time-consuming and costly. As an IVD manufacturer, ViennaLab takes care of all these requirements, as confirmed by the certificate issued by the Notified Body TÜV Süd. View this link here.
So, after selecting ViennaLab as your supplier of diagnostic products, you can sit back, relax, and diagnose.
New family member
Think big with a smaller kit configuration
Are you planning a new microbiome project? You do not have many samples to work on at the beginning?
ViennaLab has a perfect solution for you. We now offer a smaller kit configuration – 16 reactions. This attractive set-up is intended to provide an economical solution for new users to familiarize themselves with the assay and for labs with a low sample throughput.
The new configuration is already on stock!
Follow this link to view more details.

ESHG 2023 Conference
56th European Society of Human Genetics Conference is taking place in Glasgow/UK
The live event will be held from June 10 - 13, 2023.
Scottish Event Campus / United Kingdom
ViennaLab is exhibiting together with our mother company, BioVendor, at booth #410.
We are looking forward to your visit!

Frühjahrstagung 2023
Österreichische Gesellschaft für Klinische Pathologie und Molekularpathologie / Österreichische Abteilung der Internationalen Akademie für Pathologie (ÖGPath/IAP Austria)
Die Frühjahrstagung findet vom 24. bis 25. März 2023 statt.
TechGate Vienna, Donau City Wien
ViennaLab wird abermals vom österreichischen Vertriebspartner HVD Life Science Vertriebs GmbH bei der Industrieausstellung vertreten.
Wir freuen uns auf Ihren Besuch auf unserem Messestand!

NGS Contract Award
ViennaLab Diagnostics is part of the HUFIAT Consortium awarded a contract for Phase 1 to develop an end-to-end NGS solution in the field of liquid biopsy (cfDNA).
ViennaLab Diagnostics will apply their expertise in cfDNA and genetic tests and join forces with bioinformatics experts – Euformatics and AI specialists – Oncompass Medicine to work on a solution which aims to improve the field of liquid biopsy and cancer detection.
With this non-invasive approach we will develop a Pan-cancer cfDNA test to profile the tumor landscape which will allow to predict the best personalized treatment for every patient.
Follow the link to get more information on OncNGS . Please click here for the official Press Release.

Medlab Middle East 2023
Medlab Middle East 2023 is taking place as usual!
The live event will be held from February 6 - 9, 2023.
ViennaLab is exhibiting together with our mother company, BioVendor, at booth Z5.D30 and in parallel with HVD VertriebsGmbH at booth Z5.C20.
We are looking forward to your visit!
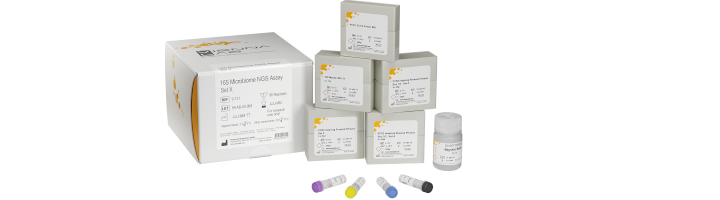
16S Microbiome NGS Assay
Check out our improved 16S Microbiome NGS Assay
We now offer an improved version of our NGS Assay with a more easier and even faster library preparation protocol bundled with data analysis for accurate species level-classification.
Improvements:
- Fast and optimized 2-step protocol minimizes set-up time
- More balanced heterogeneity spacers to enhance library complexity for improved sequencing quality
- Elimination of PhiX spike-in results in lower reagent expenses, more samples per sequencing run and therefore lower costs per sample
For more detailed information follow this link.

Season's Greetings
Thank you!
This year is coming to an end and thus we want to use this opportunity to thank our business partners, customers and distribution partners for your trust in us.
We wish you and your family happy holidays and a prosperous New Year!
ViennaLab Team

MEDICA Trade Fair 2022
Visit us at MEDICA 2022
The live event will be held from November 14 - 17, 2022 and we will be on site.
We are exhibiting together with our mother company, BioVendor, in Hall 3 A92.
We are really looking forward to meet with you again in person!

MedPhab
ViennaLab Diagnostics is part of the MedPhab consortium funded by a Horizon 2020 research grant.
MedPhab serves as Europe’s first Pilot Line dedicated to manufacturing, testing, validation and up-scaling of new photonics technologies for medical diagnostics. The purpose of MedPhab pilot production line is to accelerate the commercialisation of diagnostic devices and instruments for treatment based on photonics, and to reduce the R&D costs. pdf Read more.
Medphab is launching an Open Call for external companies developing medical products. pdf Read more.








